
When the immiscible solvents are mixed, some compounds can be drawn into the other layer if they have better solubility in that solvent. This technique is useful to separate reaction product mixtures where the desired product and the side products have different solubilities. The more dense, top layer (often the organic layer) can then be put into a separate beaker. The stopcock at the bottom of the separating funnel is opened to remove the bottom layer into another flask and the stopcock is close again to achieve complete separation of the layers. In practical organic chemistry, this extraction method of separation can be performed using a separating funnel, which holds the liquids in a way that they can separate based on their different densities. The aqueous phase forms at the bottom because water is denser, and the oil layer forms on top since it has a lower density. 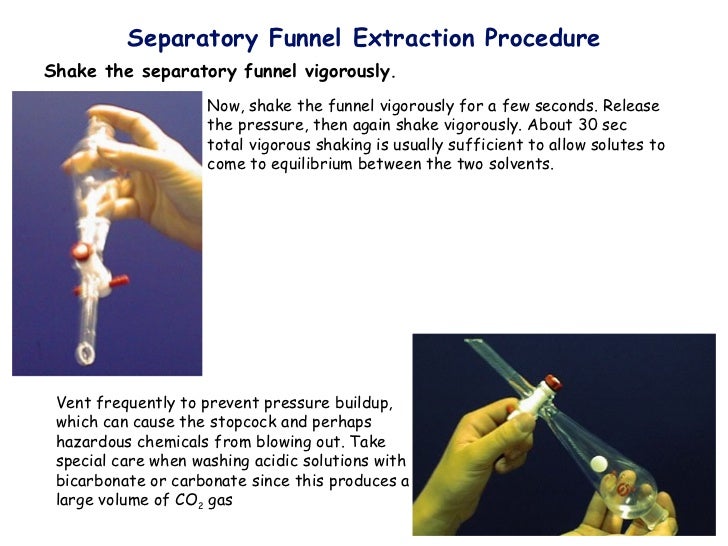
A good example of separable, immiscible liquids is water and oil. The aqueous phase contains water and other charged ions and species, whereas the organic phase contains neutral compounds in the organic solvent. These form into two phases, the organic phase, and the aqueous phase. A solvent with a higher density will be on the bottom and the lower density will be on the top. When two immiscible liquids are mixed (like oil and water) they will form two layers when put into a separating funnel due to their different densities. Extraction is a method used to isolate a chemical from a mixture based on its relative solubility in two immiscible solvents.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
